Abstract
Background: The therapeutic mechanism of repetitive transcranial magnetic stimulation (rTMS) for treatment-resistant depression (TRD) may involve modulation of γ-aminobutyric acid (GABA) levels. We used proton magnetic resonance spectroscopy (MRS) to assess changes in GABA levels at the site of rTMS in the left dorsolateral prefrontal cortex (DLPFC).
Methods: In 26 adults with TRD, we used Mescher–Garwood point-resolved spectroscopy (MEGA-PRESS) spectral-editing MRS to measure GABA in the left DLPFC before and after standard clinical treatment with rTMS. All participants but 1 were medicated, including 12 patients on GABA agonist agents.
Results: Mean GABA in the DLPFC increased 10.0% (p = 0.017) post-rTMS in the overall sample. As well, GABA increased significantly in rTMS responders (n = 12; 23.6%, p = 0.015) but not in nonresponders (n = 14; 4.1%, p = not significant). Changes in GABA were not significantly affected by GABAergic agonists, but clinical response was less frequent (p = 0.005) and weaker (p = 0.035) in the 12 participants who were receiving GABA agonists concomitant with rTMS treatment.
Limitations: This study had an open-label design in a population receiving naturalistic treatment.
Conclusion: Treatment using rTMS was associated with increases in GABA levels at the stimulation site in the left DLPFC, and the degree of GABA change was related to clinical improvement. Participants receiving concomitant treatment with a GABA agonist were less likely to respond to rTMS. These findings were consistent with earlier studies showing the effects of rTMS on GABA levels and support a GABAergic model of depression.
Introduction
Up to 44% of patients with major depressive disorder (MDD) experience treatment-resistant depression (TRD): illness that fails to respond to multiple medication trials.1 Among patients with MDD, those with TRD have appreciably higher morbidity and mortality.2,3 Repetitive transcranial magnetic stimulation (rTMS) is effective for TRD, resulting in remission in approximately 35% of patients and appreciable improvement in about another 30%.4 Research interest remains high in elucidating the mechanism of action of rTMS to increase its efficacy for TRD and other conditions.
It has been hypothesized that the mechanism of action of rTMS, as well as other antidepressant treatment modalities, involves central metabolism of γ-aminobutyric acid (GABA). Increasing evidence implicates GABA and GABAergic receptors in MDD, TRD and their treatment.5–8 Compared with healthy controls, GABA levels measured in plasma, cerebrospinal fluid (CSF) and in vivo brain through proton magnetic resonance spectroscopy (MRS) are reduced in people with MDD and TRD.9 Moreover, in some (but not all) MRS studies, cortical GABA increased following treatment with selective serotonin reuptake inhibitors,9–11 electroconvulsive therapy,12 cognitive behavioural therapy13 or intravenous ketamine.14 Thus, depleted GABA has been widely observed anatomically in MDD, and GABA has been shown to increase following a wide range of MDD treatment modalities.
Potential GABA changes associated with rTMS have not been extensively explored, but a recent naturalistic clinical study of TRD15 using J-difference-edited MRS optimized for GABA quantitation16,17 measured GABA levels in the midline pregenual anterior cingulate cortex (pACC) following 10 Hz rTMS applied to the left dorsolateral prefrontal cortex (DLPFC).18 In response to rTMS, GABA rose in all patients, more in clinical responders than in nonresponders. The pACC and neighbouring anterior cingulate subregions are important in the pathogenesis of MDD.19,20 It is not known whether similar GABAergic effects of rTMS are seen at the primary treatment target in the left DLPFC; neuroimaging investigations have in fact reported widespread changes in metabolism or perfusion following rTMS, without any significant change at the treatment target in the left DLPFC.21–23 Dysregulation of GABA in the DLPFC has been reported in patients with MDD, marked by loss of GABAergic interneurons, 24 abnormal subunit composition of GABAA receptors25,26 and lower GABA levels than in healthy controls on MRS.27 While a negative correlation between baseline DLPFC GABA and treatment response to rTMS has been reported,28 and theta-burst rTMS has been shown to decrease the ratio of GABA to glutamate + glutamine (Glx) in the left DLPFC in healthy participants,29 it is not known what GABAergic changes would be seen at the stimulation site in patients with TRD following rTMS.
The primary aim of the present study was to examine whether rTMS administered to the left DLPFC was associated with increased GABA at the stimulation site, and whether these changes differed between responders and nonresponders to rTMS, as reported by Dubin and colleagues for the pACC.15 The present investigation was an open-label naturalistic trial that enabled us to determine whether a GABA increase in the DLPFC could be observed under naturalistic treatment conditions, in which patients were allowed to continue concomitant psychotropic medication. Because 12 of 26 patients were taking GABAergic agonist drugs (e.g., benzodiazepines), we also explored whether the use this class of medications was related to clinical response and/or GABA levels.
Methods
Study design
Participants were patients with MDD who were referred to the UCLA TMS Clinical and Research Service and who underwent a 6-week acute course of rTMS treatment for TRD. Clinical symptoms were assessed weekly over the course of treatment. We performed MRS scans for each participant at baseline and again at completion of the course of treatment (30 sessions). The analysis sample included the first 26 patients who had both pre- and post-rTMS GABA MRS scans that passed quality-control criteria (see below). We examined baseline and post-rTMS GABA levels in relationship to clinical outcome in terms of percent change on the 30-item Inventory of Depressive Symptoms (IDS-SR30) total score.30,31 Although the Hamilton Depression Rating Scale (HAM-D) is more commonly used in clinical trials, the IDS-SR30 captures all 9 DSM-IV criterion symptom domains and atypical features for MDD, it provides reliable measures of symptom severity and change, it allows for rapid appraisal of clinically relevant symptom features, it is sensitive to symptom change for multiple treatment modalities, and it is comparable in performance to the HAM-D.32 Baseline, interim-treatment and final post-treatment scores on the IDS-SR30 were available for all patients, but only baseline and interim-treatment HAM-D scores were available for all patients. Similar to Dubin and colleagues,15 clinical response was defined as a ≥ 30% reduction in IDS-SR30 score after rTMS treatment.30 This is equivalent to a HAM-D reduction of 28.6%.33 In addition to the 30% responder criterion, we examined post-rTMS change in the principal study imaging outcome (left DLPFC GABA) for responders and nonresponders using the alternative criteria of 25%, 35%, 40% and 50% reduction in IDS-SR30 score.
Participants
Participants comprised 26 adults (12 male, 13 female, 1 other; age 38.4 ± 13.8 years [mean ± 1 standard deviation]; education 16.0 ± 3.5 years) with a primary DSM-IV diagnosis of MDD confirmed with the Mini-International Neuropsychiatric Interview (MINI)34 who were recruited from the UCLA TMS Clinical and Research Service, where they had been referred with at least 2 failed trials of antidepressant medication. Informed consent was obtained from all participants before research procedures. All procedures were approved by the UCLA Office of the Human Research Protection Program Medical Institutional Review Board. Participants were excluded if they were mentally or legally incapacitated and unable to give informed consent; were pregnant; had epilepsy, dementia, a history of repetitive or severe head trauma, or tumours in the central nervous system; or had an intracranial implant such as a cochlear prosthesis, electrodes, aneurysm clips or coils, cardiac pacemaker, vagus nerve stimulator or deep brain stimulator or other devices or objects contraindicated for rTMS and/or MRI. As is typical of TRD clinical samples, all but 1 participant was being treated with 1 or more psychopharmacologic agents concomitant with rTMS. Patients were encouraged to continue their pre-rTMS medications during treatment but were not prohibited from medication changes as prescribed by their outpatient psychiatrist. Of special relevance, 12 participants were being treated with 1 or more GABAergic agonist agents (principally benzodiazepines, but also possibly non-benzodiazepine anticonvulsants/mood stabilizers, hypnotics or sedatives) and 14 patients were not receiving such drugs concurrent with the rTMS regimen. The classes of psychoactive medications given to each patient at time of rTMS are listed in Appendix 1, available at jpn.ca/180230-a1.
Transcranial magnetic stimulation treatment
All participants began 5-day-per-week, 6-week rTMS treatment. Individual sessions consisted of 3000 pulses (10 Hz, 4 s duration, 26 s interval; for 1 patient, right DLPFC) administered with either the NeuroStar TMS System (Neuronetics, Inc.) or the Magstim Rapid2 Therapy System (Magstim, Inc.). The stimulating magnet was placed over the left DLPFC using the Beam F3 method.35 Treatment was initiated at 80% to 90% motor threshold (MT) and advanced as rapidly as tolerated to an intensity of 100% to 120% MT for the majority of patients (Appendix 1). Physicians were allowed to adjust the stimulation frequency, intensity and number of pulses per session as needed based on tolerability and response to treatment. After the 15th treatment session, sequential bilateral treatment could be performed, adding 1 Hz treatment to the right DLPFC for nonresponse to left unilateral treatment (19 participants). One participant was unable to tolerate left unilateral treatment and received stimulation solely over the right DLPFC; another was switched from left to right DLPFC stimulation starting with the fifth treatment (Appendix 1).
MRI procedures
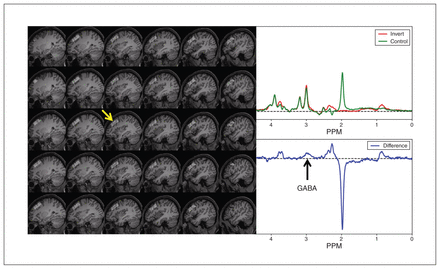
We performed neuroimaging at 2 time points — baseline and post-treatment — on a 3 T Siemens Prisma with 64-channel phased-array head coil at the UCLA Ahmanson–Lovelace Brain Mapping Center. Imaging included whole-brain sagittal structural MRI (magnetization prepared rapid acquisition gradient echo [MPRAGE], voxels 0.8 × 0.8 × 0.8 mm3) and water-suppressed single-voxel 1H MRS (repetition time 2000 ms, echo time 68 ms, 192 excitations) of the left DLPFC stimulation target area using the Mescher–Garwood point-resolved spectroscopy (MEGA-PRESS)36 spectral-editing technique for GABA. This pulse sequence was an investigational “works-in-progress” package provided by Siemens. The DLPFC MRS voxel was prescribed (Fig. 1) as in studies by Kegeles and colleagues37 and Simpson and colleagues,38 with modifications. An MRI-opaque vitamin E capsule with its long axis oriented roughly parallel to the anticipated rostral–caudal direction of the gyrus was firmly affixed with surgitape to a cloth electroencephalogram cap at the F3 stimulation site. This method facilitated placement of the MRS acquisition volume in the cortex deep to the selected electro-encephalogram scalp electrode site. After MPRAGE was acquired, it was resliced online using the 3D reconstruction utility at the Prisma console into 3 orthogonal whole-brain volumes. The first was an axial–oblique volume parallel to the longitudinal axis of the left hippocampus. The second was a coronal–oblique volume normal to the hippocampal volume. The third volume, sagittal–oblique, was constructed as follows. In the coronal– oblique volume, the slice that contained the anterior commissure was identified and a line was drawn from the midpoint of the commissure to the centre of the vitamin E capsule at F3. In the coronal–oblique view, the sagittal plane was rotated inferolaterally to match this angle, and this determined the orientation of the third volume. The MRS voxel thus had faces parallel to each of these MRI volumes and was positioned in the cortex directly under the vitamin E capsule, with care taken to ensure that no part of the voxel exited the brain. The voxel measured an initial 30 × 20 × 10 mm3, adjusted in size and orientation to maximize local DLPFC grey matter content. We acquired an identical, non-water-suppressed scan (8 excitations) immediately afterward from the same site.
Meshcher–Garwood point resolved spectroscopy (MEGA-PRESS) voxel position and representative spectra. Left: series of sagittal T1-weighted MRI sections of the brain of a representative patient with treatment-resistant depression, showing the position of the 1H magnetic resonance spectroscopy (MRS) MEGA-PRESS (repetition time 2000 ms, echo time 68 ms) acquisition volume (voxel; pale areas, yellow arrow) in the left middle frontal cortex (dorsolateral prefrontal cortex). Upper right: averaged MEGA-PRESS spectra acquired on alternate scans with (invert, red) and without (control, green) the frequency-selective inversion pulse that avoids excitation of the γ-aminobutyric acid (GABA) C3 peak at 1.9 ppm. Lower right: edited spectrum (difference) generated by subtracting the invert from the control MEGA-PRESS spectrum. This editing operation yields a spectrum in which the GABA resonance at 3.0 ppm (obscured in conventional PRESS MRS by the much larger overlying creatine + phosphocreatine peak) can be quantified, because it appears in relative isolation from other metabolite signals.
We obtained GABA levels from LCModel fitting of the edit spectrum, which included adjustment for the (possibly variable) volume of the MRS acquisition voxel. Spectra with obvious artifacts (head motion, lipid contamination, inadequate water suppression, eddy currents not removed by LCModel) or poor quality (full-width at half-maximum > 0.15 ppm, signal-to-noise ratio < 3) were rejected. Individual metabolite signals with Cramer–Rao lower bounds > 20% were rejected. The GABA levels for the edited spectrum were normalized to the unsuppressed water signal of the noninverted spectrum. The MPRAGE was tissue-segmented into grey matter, white matter and CSF subvolumes using FSL FAST (http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FAST). Using software custom-written by one of the authors (J.R.A.), we determined MRS voxel tissue composition by coregistering the MRS voxel to these subvolumes. This software also implemented CSF correction of GABA levels.39 Water-referenced, CSF-corrected GABA levels were expressed in institutional units (IU).
Statistical analyses
We calculated group-mean clinical outcomes of treatment for the responder and nonresponder subgroups. We used a paired t test to compare post- to pre-rTMS IDS-SR30 scores to determine whether there was a significant effect of treatment on the severity of depressive symptoms for the overall sample. Based on previous clinical trials,4,15 we expected a priori that rTMS treatment would reduce IDS-SR30 scores. We next examined the major potential confound for effects on GABA levels by evaluating whether group-mean tissue composition (grey matter, white matter, CSF) of the MRS acquisition voxel varied with GABA at baseline or changed pre- to post-rTMS. For the primary aim, we also had a priori hypotheses that left DLPFC GABA levels would increase post-rTMS, particularly in the responder subgroup. For extra precaution, however, we controlled for multiple comparisons by testing for the primary aim using an omnibus repeated-measures analysis of variance, which examined the within-patient main effect of rTMS on left DLPFC GABA levels, using the interaction terms of response status, concomitant use of GABA agonists and stimulation site (left hemisphere, right hemisphere or sequential bilateral). We performed post hoc t tests on subgroups as appropriate in the case of significant interaction terms. Drawing on the exploratory findings of Dubin and colleagues, 15 we further tested whether there were significant Pearson correlations between Glx and GABA in the left DLPFC at baseline and after rTMS. Finally, we compared the number of responders in the patient subsamples taking GABA agonist drugs using a Fisher exact test and used an independent t test to compare these subgroups for differences in the mean post-versus pre-rTMS percent changes in IDS-SR30 score. We performed statistical analyses using SPSS 25 (SPSS Inc.). Although directional hypotheses would have permitted 1-tailed testing in some cases, we kept the criterion for significance at p < 0.05 (2-tailed) for all analyses to be uniform and conservative.
Results
Clinical outcome: effects of rTMS treatment on depressive symptoms
We observed a significant effect of rTMS treatment on depressive symptoms, with a group-mean final post-rTMS IDS-SR30 score (Table 1) of 33.5 ± 18.0 (range 2–71), representing a mean IDS-SR30 change of −32.3% ± 37.2% (−96.2% to 22.7%; paired t test t25 = −3.8, p = 0.001). Twelve patients with TRD were classified as responders, and 14 as non-responders. Using a 30% reduction in HAM-D score and last observation carried forward as an alternative definition, there were 11 responders and 15 non-responders, nearly the same as for the IDS-SR30 results. Across patients, IDS-SR30 change did not correlate significantly with the rTMS stimulation intensity (percent resting motor threshold) used for the majority of sessions during treatment (Pearson r = −0.087, p = 0.68). We found no significant differences between patients treated with the Neurostar and Magstim instruments in terms of mean IDS-SR30 change (t = 0.7, p = 0.51) or number of responders (Fisher exact test p = 0.68).
Demographic and clinical characteristics of study participants
MRS voxel tissue composition
For the overall sample, the grey matter content of the left DLPFC MRS voxel (Table 2) ranged from about 30% to 60% for both pre- and post-rTMS scans. These were favourable values, considering the relatively large MRS voxels required for GABA MEGA-PRESS acquisitions. White matter content ranged from 20% to 65%, and CSF content ranged from 6% to 30%. We found no significant correlations at baseline between GABA levels and MRS voxel tissue composition. We found no significant post/pre differences in voxel tissue composition.
MRS GABA levels in left DLPFC before and after TMS
Effects of rTMS treatment on MRS GABA levels
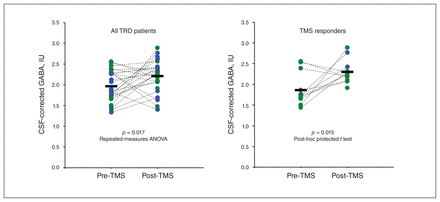
Across the total sample, we observed a mean 10.0% increase in left DLPFC GABA levels after rTMS treatment compared with baseline (2.0 ± 0.4 IU pre-rTMS v. 2.2 ± 0.4 IU post-rTMS; repeated-measures analysis of variance F1,20 = 6.8, p = 0.017; Fig. 2). We found no significant main effect of responder status, but we did observe a significant rTMS × response interaction (F1,20 = 5.7, p = 0.027). We found no significant interactions involving the use of GABA agonists, or left- or right-sided rTMS. Post-hoc t tests revealed that within the responder subsample, mean left DLPFC GABA levels increased 23.6% after rTMS (2.3 ± 0.3 IU v. 1.9 ± 0.4 IU; t11 = −2.9, p = 0.015; Fig. 2). Among the 14 nonresponders, DLPFC GABA levels increased 4.1% from post- to pre-rTMS (2.1 ± 0.4 IU v. 2.0 ± 0.4 IU; not significant). We also observed a significant increase in left DLPFC GABA levels for responders but not for nonresponders using the alternative responder criteria of 25% (p = 0.007), 35% (p = 0.007) and 40% (p = 0.033) decreases in IDS-SR30 post-rTMS. Using a 50% drop in IDS-SR30 as a criterion, we observed a trend-level decrease in GABA (p = 0.07). Left DLPFC GABA change did not correlate significantly with change in IDS-SR30 score (Pearson r = −0.31, p = 0.13) or with the rTMS intensity (% resting MT) used for the majority of sessions during treatment (Pearson r = 0.097, p = 0.65). We observed no significant difference between the Neurostar and MagStim instruments in left DLPFC GABA change (t = −1.4, p = 0.18).
γ-Aminobutyric acid (GABA) levels in the left dorsolateral prefrontal cortex. Left: pre- and post–repetitive transcranial magnetic stimulation (rTMS) cerebrospinal fluid (CSF)–corrected levels of GABA in the left dorsolateral prefrontal cortex of the full sample of patients with treatment-resistant depression (TRD) (n = 26). Each participant is indicated by a pre/post pair of dots connected by a dashed black line. Blue dots are for patients who took GABAergic agonist medication concurrent with the rTMS regimen; green dots are for patients who did not take GABA medication; black horizontal bars denote group means. For the overall sample, GABA increased by a mean of 10.0% after rTMS (F1,20 = 6.8, p = 0.017, repeated-measures analysis of variance). Right: the same analysis for rTMS responders only (n = 12). For this subsample, GABA increased a by mean of 23.6% after rTMS (t11 = −2.9, p = 0.01, post hoc protected t test). For the rTMS nonresponders (n = 14), 10 of whom were taking GABA agonists, GABA increased by a mean of only 4.1% after rTMS (p = NS). Responder status based on ≥ 30% reduction in 30-item Inventory of Depressive Symptomsscore after rTMS treatment.30 IU = institutional units; NS = not significant.
DLPFC MRS Glx versus GABA correlations before and after rTMS treatment
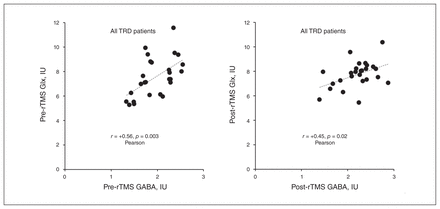
At baseline across the total sample, we found a positive correlation between left DLPFC Glx and left DLPFC GABA (Pearson r = 0.56, p = 0.003; Fig. 3). After rTMS treatment, this correlation was weaker but still significant (r = 0.45, p = 0.02).
Glutamate + glutamine (Glx) versus γ-aminobutyric acid (GABA) levels in left dorsolateral prefrontal cortex before and after repetitive transcranial magnetic stimulation (rTMS). Left: pre-rTMS cerebrospinal fluid (CSF)–corrected levels of Glx versus GABA in the left dorsolateral prefrontal cortex of the full sample of patients with treatment-resistant depression (TRD) (n = 26). Each participant is indicated by a dot. The dashed line represents the least squares fit of the data. Glx increased with increasing GABA (r = +0.56, p = 0.003, Pearson). Right: the same analysis for post-rTMS. Though less strongly, Glx still increased significantly with increasing GABA (r = +0.45, p = 0.02). IU = institutional units.
Effects of GABA agonist drugs on clinical response
Of the 12 patients taking GABAergic agonist medications, 2 were rTMS responders and 10 were rTMS nonresponders. Of the 14 patients not taking GABA agonist medications, 10 were rTMS responders and 4 were rTMS nonresponders. This between-group difference in number of responders was statistically significant (Fisher exact test p = 0.008). Consistent with this, the mean post-rTMS drop in IDS-SR30 was 30.2% greater for the subgroup that was not taking GABA agonists than for the subgroup that was taking these agents (independent t test t24 = −2.2, p = 0.035).
Discussion
To our knowledge, this was the first MRS study to measure effects of rTMS on GABA in the left DLPFC stimulation site in a clinical TRD sample. We found that while GABA increased in the overall TRD sample, there was a significant interaction with treatment response, such that the increase in GABA was significant only for the subgroup that responded to rTMS treatment. The increase in GABA was not affected by concomitant treatment with GABA agonist medication, although the rate and degree of treatment response were lower in patients taking such medication. The first finding suggests that rTMS affects GABAergic metabolism in the brain and that the DLPFC itself — as opposed to only remote sites — shows the metabolic effects of rTMS. These findings also suggest that greater GABA increases may attend superior clinical response.
Our findings in the DLPFC closely resemble those of Dubin and colleagues15 in the pACC, demonstrating post-rTMS changes in GABA in both rTMS responders and nonresponders, but significant changes only in the responder subgroup. Both investigations used a spectral-editing MRS protocol to optimize the detection of GABA. The present results suggest that the DLPFC stimulation site itself is metabolically affected by rTMS, in addition to conducting effects to other remote brain sites. These studies add to a growing body of evidence that GABA levels increase in response to a variety of treatments for MDD, including selective serotonin reuptake inhibitors,10 electroconvulsive therapy12 and ketamine.14 Other studies failed to find significant post-treatment changes in GABA.40,41 Discrepancies may be due to differences in technique, acquisition site, heterogeneity of illness, severity of illness42 or, as seen here, use of GABAergic medications. A recent meta-analysis9 supports a GABAergic model of MDD,43–45 finding evidence across plasma, CSF and MRS studies for diminished GABA levels in depressed patients that revert toward control levels in euthymia. Post-mortem studies in patients with MDD compared with healthy controls demonstrate reduced calbindin immunoreactive GABAergic neurons in prefrontal and occipital cortices, 24,46 reduced calbindin and calretin immunolabelled GABAergic neurons in the auditory cortex47 and decreased expression of somatostatin (an inhibitory neuropeptide) in GABA+/calbindin+ inhibitory interneurons48 at the mRNA and precursor peptide levels in the subgenual anterior cingulate cortex49 and DLPFC.50 The present investigation found a greater post-rTMS increase of GABA in responders than in nonresponders. Using conventional PRESS MRS not optimized for measuring GABA, Baeken and colleagues28 detected a negative correlation of DLPFC GABA with treatment response, also indirectly implicating GABA in rTMS treatment mechanisms. The MEGA-PRESS pulse sequence of our study was optimized for GABA, and our results further support a role for GABA in rTMS response.
In an exploratory analysis at pre-rTMS baseline, Dubin and colleagues15 found a significant positive correlation between Glx and GABA in the pACC for their 12 patients with severe MDD (HAM-D > 27). This correlation was no longer significant after the rTMS trial. In their 11 patients with moderate MDD (HAM-D ≤ 27), there was no significant correlation before or after rTMS. By the same criteria, the present sample had 20 moderate and only 6 severe MDD patients; we deemed the latter to be too few to stratify by severity. Therefore, we plotted Glx versus GABA for the entire patient sample, which resulted in significant positive correlations both before and after rTMS. The rTMS-induced decoupling of Glx and GABA metabolism supported by the results from Dubin and colleagues15 may be specific to the pACC.
Finally, we found that the use of GABA agonists was not associated with changes in GABA levels, but was associated with inferior treatment response. While these findings may appear contradictory, GABA agonists exert specific effects at various GABAA receptors in the brain that may or may not translate to changes in the gross tissue levels of GABA measured by MRS. It is conceivable that these agents may impede rTMS therapeutic action without affecting metabolic GABA levels per se. Clinical studies have demonstrated that benzodiazepines elevate the rTMS resting motor threshold in patients with MDD and in other participants.51,52 Preclinical studies demonstrate downregulation of α-1 GABAA receptor subunit mRNA53–55 and decreased α-1 GABAA receptor subunit polypeptide54,56,57 in the cortex of rats subjected to chronic administration of benzodiazepines. As well, GABA agonists could influence rTMS response via voltage-dependent inhibition of Na+ channels, 58 or reduction of voltage-dependent Ca2++ currents.59 Since submission of this report, the finding of lower rTMS efficacy for patients with MDD receiving GABA agonist cotreatment has been replicated in a larger independent sample by our group60 and at another centre.61 Neither of these other studies acquired MRS GABA. Further investigation in larger groups is needed to differentiate the impact of comorbid disorders such as anxiety or insomnia from that of GABAergic medications as described above on rTMS clinical outcome and determine whether discontinuing GABA agonists is warranted during rTMS regimens.
Limitations
Limitations of this naturalistic study included small sample size; open-label design; lack of sham rTMS control; and some heterogeneity across patients in medication use (including GABAergic agonists), medication changes during treatment, treatment parameters (hemisphere and intensity of stimulation) and psychiatric comorbidities. Findings should be considered exploratory and need to be replicated in larger future randomized controlled trials. The heterogeneity introduced by use of GABA agonists and different treatment sites was mitigated to some extent by the use of covariates in the statistical model. Although within-patient stimulation intensity was varied to optimize symptom reduction, between-patient variation in stimulation intensity did not influence GABA or clinical response. Furthermore, the naturalistic design enabled us to state that rTMS effects on GABA were sufficiently robust to be observed under actual conditions in a clinical setting. The stimulation magnet was positioned over the left DLPFC using the Beam F3 technique according to our usual clinical practice, but future studies may profit from MRI-guided neuronavigation for this purpose.
Conclusion
Present findings further support the notion that regional brain GABA levels increase at the site of stimulation in response to successful rTMS treatment of MDD.10,11,14,15 This suggests that the DLPFC is not simply a “pass through” zone, but a region that undergoes metabolic changes during rTMS. Use of GABAergic agonists concurrent with rTMS may be detrimental to clinical recovery from TRD.
Acknowledgements
The authors are grateful to Lawrence S. Kegeles, MD, PhD, of the New York State Psychiatric Institute for details of the prescription of the DLPFC MRS voxel. The authors thank Sinyeob Ahn, PhD, of Siemens Healthcare, San Francisco, for writing the MEGA-PRESS works-in-progress pulse sequence.
Footnotes
Competing interests: I. Cook reports a grant from NeoSync and earns equity interest from Los Angeles TMS Institute Inc., Neuro- Sigma Inc., HeartCloud Inc., and BrainCloud Corporation, outside the submitted work. A. Leuchter reports grants from the National Institutes from Health, Neuronetics, NeuroSigma Inc., the US Department of Defense, and the CHDI Foundation. He is a consultant and stockholder of NeoSync Inc., a consultant with Ionis Pharmaceuticals and EIMindA, and earns equity from Brain Biomarker Analytics LCC. No other competing interests declared.
Contributors: J. Levitt, J. O’Neill and A. Leuchter designed the study. J. Levitt, G. Kalender, J. O’Neill, J. Diaz, I. Cook, N. Ginder, D. Krantz, M. Minzenberg, N. Vince-Cruz, L. Nguyen and A. Leuchter acquired the data, which J. Levitt, G. Kalender J. O’Neill, M. Minzenberg, J. Alger and A. Leuchter analyzed. J. Levitt, J. O’Neill, J. Diaz, N. Ginder, D. Krantz, M. Minzenberg, N. Vince-Cruz, L. Nguyen, J. Alger and A. Leuchter wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received November 18, 2018.
- Revision received February 22, 2019.
- Accepted March 4, 2019.